header
News
Establishing a Scientific Roadmap to Protect Public Health Amid Climate Threats
- WRITER 학무부총장실

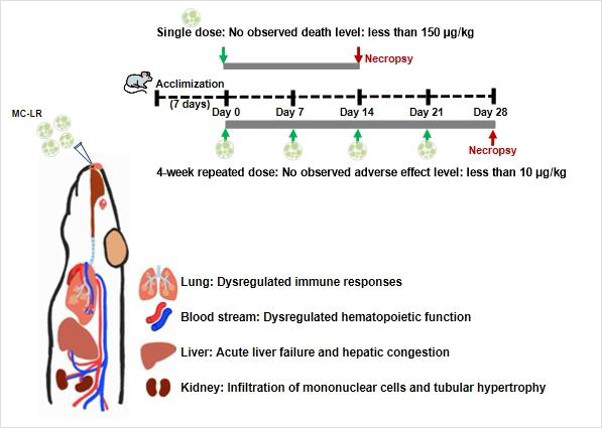
A research team led by Professor Eun-Jung Park of the College of Medicine identifies the respiratory risks of “microcystin” found in green algae and proposes safety guidelines for the public. Utilizing experimental animals and a 3D reconstructed human airway model, the team (including Professors Jin-Bae Kim, Cheon Woong Choi, and Moon-hyung Lee) derived Maximum Non-Lethal Dose (MNLD), the No-Observed-Adverse-Effect Level (NOAEL), and the Tolerable Daily Intake (TDI) to safeguard health from green algae risks.
“While concerns have been raised regarding the generation of aerosols and fine dust—and the subsequent potential for inhalation—alongside the occurrence of green algae blooms, there have been no established standards for assessing toxicity levels or human safety guidelines.”
- Professor Eun-Jung Park, College of Medicine
Microcystin found in green algae was found to remain in liver tissue for up to one week after entering the body through the respiratory system. Professor Park emphasized the importance of this oversight, stating, “Total exposure must be strictly managed to stay below 10 μg, taking all potential exposure routes into account.”
The acceleration of global warming is leading to extensive problems such as extreme climate events (heatwaves, heavy rains, droughts, wildfires) due to the rise in average global temperatures, as well as rising sea levels, glacial retreat, ocean acidification, food and water shortages, and ecosystem destruction. In addition, it increases the frequency and severity of green algae blooms. Across South Korea’s major waterways, including the Four Major Rivers, toxic green algae blooms have become a recurring summer phenomenon, leaving the water looking as though it has been stained with vivid green paint. Recently, health problems among residents near the Nakdong River have become a major environmental issue, and green algae is emerging as a serious public health problem threatening national health alongside climate change. The research team led by Professor Eun-Jung Park of the College of Medicine conducted a study related to the toxicity of green algae. We heard from Professor Park regarding the results of this research. <editor's note>
Hepatic congestion found in all experimental animals upon repeated exposure, resulting in death or near-death
Q. While the risks of green algae have been discussed previously, what are the specific details of this study?
Existing research has primarily focused on toxicity associated with consuming contaminated drinking water, which led to the establishment of safety standards for water consumption. However, green algae blooms also generate aerosols in the form of water mist or fine droplets, as well as fine dust. These particles can disperse into the air and be inhaled through the respiratory system, such as the nose and lungs, yet no standards existed regarding toxicity or human safety for such inhalation. We recognized the necessity of proposing guidelines based on scientific evidence to protect the health of residents in areas affected by green algae blooms.
Utilizing experimental animals and 3D artificial nasal models, we presented the Maximum Non-Lethal Dose (MNLD), the No-Observed-Adverse-Effect Level (NOAEL), and the Tolerable Daily Intake (TDI) for microcystin. The MNLD represents the highest concentration at which all experimental subjects survive, while the NOAEL is the concentration at which no harmful effects on humans are judged to occur. The TDI is the maximum daily dose of a specific substance—such as an environmental contaminant—to which a person can be exposed every day for a lifetime without an appreciable health risk.
Q. A unique aspect of this research is that it assumes toxins enter the body through the respiratory system (nose, lungs, etc.). What were the findings?
First, to determine the possibility of entry through the respiratory system, we examined the size of aerosols containing microcystin. Most of the aerosols were 5 μm (micrometers) or smaller, a size that allows them to be inhaled through the respiratory tract. Using a 3D reconstructed human airway model that mimics the human nasal cavity, we observed whether inhaled microcystin could pass through the nasal epithelium. The results showed that microcystin increased mucus secretion and penetrated the nasal epithelium just three hours after exposure. We concluded that microcystin has the potential to migrate to other tissues via the nasal epithelium.
In the next stage, we confirmed acute toxicity levels. Microcystin was administered once to the nasal cavities of male mice at doses of 30, 150, and 300 μg/kg. Deaths were observed in the groups exposed to 150 and 300 μg/kg. This indicates that the MNLD for a single respiratory exposure to microcystin is 150 μg/kg or less.
To derive safe concentrations for repeated exposure, we administered doses of 10, 50, and 100 μg/kg to both male and female mice once a week for a total of four weeks. Approximately two hours after the second administration of the 100 μg/kg dose, animal deaths began to occur. Notably, female mice tended to be more sensitive to microcystin than males. While we initially predicted lesions in the lung tissue during the study design, the actual lung lesions were minimal. However, hepatic congestion was observed in all experimental animals that died or were near death.
Cell damage and “lamellar bodies” accumulate under stress, confirming microcystin’s impact on liver cells
Q. What is the significance of hepatic congestion, and were there any other observed changes?
Hepatic congestion refers to the accumulation of blood in the liver. The livers of the mice that died or were near death were severely damaged. We concluded that the accumulation of microcystin in the liver triggered abnormal reactions. Specifically, the concentration of C-reactive protein, an acute inflammatory marker primarily produced in the liver, also increased significantly within the lungs. Blood biochemical indicators related to hepatotoxicity also showed a marked increase. In the liver tissue of mice exposed to 10 μg, we observed necrotic cell damage, structures similar to lamellar bodies, lipid droplets, and collagen fibers.
Lamellar bodies accumulate abnormally when cells are damaged or under stress in specific situations. The discovery of these structures in liver tissue signifies that microcystin caused issues with the internal structure of liver cells, particularly regarding lipid metabolism and intracellular substance transport.
Q. The causality between hepatic congestion and mortality seems critical.
The research team investigated whether there was a direct causal relationship between microcystin inhalation, hepatic congestion, and the death of the experimental animals. We examined changes in liver tissue after single and double administrations of 100 μg of microcystin. The results showed a distinct increase in microcystin levels in liver tissue analyzed two hours after the second administration, while microcystin from the first administration remained in the liver for up to one week.
We analyzed the gene changes within the liver tissue using microarray technology. The expression of activating transcription factor 3 and nuclear receptor subfamily 4–core regulatory genes that activate when cells respond to stress or damage–increased most significantly two hours after the second exposure. In contrast, the expression of microRNA 122, which helps maintain the normal function of liver cells, decreased sharply. This indicates that the liver cells underwent severe stress within a short period of time.
In liver tissue retrieved one week after the initial exposure, we observed an increased expression of enzymes and transporters essential for processing toxic substances and regulating metabolism in the liver and kidneys, such as deiodinase 1, cytochrome P450, and solute carrier. These changes signify that the liver is enhancing its own detoxification capacity, demonstrating that the microcystin accumulated in the liver continuously stimulates the body’s internal detoxification system. Based on all derived results, we established a total allowable exposure limit for microcystin in humans, concluding that it must be managed below 10 μg, encompassing all potential exposure routes.

Addressing emerging risks through scientific research and response
Q. You have demonstrated human toxicity through animal testing, but there are differing opinions on whether these results can be directly applied to humans. In South Korea’s humidifier disinfectant case, for instance, many remember that animal testing results were not initially recognized in legal judgements. What are your thoughts on this?
I am frequently asked this question, but there is no simple answer beyond stating that we cannot conduct experiments on humans, nor can we rely solely on assumptions without verifying toxicity. Personally, I have concerns. There is a global trend in research toward emphasizing animal welfare and utilizing cells, artificial organs, or AI. Yet, all current regulations are based on animal testing. At a time when even animal testing results are sometimes contested, I worry whether values predicted by alternative testing methods will be accepted into the regulations meant to guarantee our safety.
Q. You were recently appointed as the Associate Editor of Toxicology and Applied Pharmacology, one of the oldest and most prestigious journals in the field. You are only the second Korean to hold this position; could you share the process and your thoughts on this appointment?
As an entirely domestic-trained researcher who started my research career relatively late, this was completely unexpected. Even six years ago, when I was recommended as an editorial board member for this journal, I felt I had achieved more than I ever anticipated. Consequently, whenever I was asked to review a paper, I adjusted my schedule to ensure I completed the review diligently within the deadline. I was told that the research results I accumulated during this period, combined with the trust of my academic colleagues, were decisive in this appointment. In particular, Dr. Anna Shvedova, a world-renowned authority in the field of nano-toxicity, strongly recommended me for the position.
I formed a connection with Dr. Shvedova during my time as a visiting researcher at the National Institute for Occupational Safety and Health (NIOSH) in the United States. My six-month stay was too short to derive significant scientific results like a research paper, and due to institutional regulations, visiting researchers were only allowed in the lab for 12 hours a day. Since my experiments required 24-hour monitoring, I requested a time release, which was granted with Dr. Shredova’s help. This connection led to a lasting relationship of mutual support. This appointment as Associate Editor feels like a form of encouragement from my fellow researchers for the long hours I spent in the laboratory.
Q. During the COVID-19 pandemic, you were the first to warn of the dangers of spray disinfection, and you continue to research the safe use of household chemical products that can be inhaled. What is the ultimate goal of your research?
I believe that to lose one’s health is to lose everything. A nation can only be healthy if its citizens are healthy, and for that to happen, we need minimum guidelines to protect people from hazardous substances. In the research world, AI and robotics are currently the mainstream. Many researchers wonder if they should also move into these fields.
The field of toxicology that I study is a representative area of “undone science” (science that is left unstudied or ignored). It is a very difficult world for researchers in such fields to maintain their research careers. I sometimes fear how significant the practical limitations I will face in the future might be. However, for the remainder of my career, I intend to do my best to continue ensuring that the field of “undone science” is not ignored and continues to be sustained.
※ The lead image for this article was generated using AI.
※ Related information
- Professor Eun-Jung Park’s Researcher Profile
https://professor.khu.ac.kr/sProf/E&BA5B
- Research Paper
https://www.sciencedirect.com/science/article/pii/S0147651325017063