header
News
"Professor Kwangwoo Kim's Research Team at the Department of Biology Unlocks the Secrets of Lupus Genes"
DATE 2026-01-19 16:20:25.0
- WRITER 학무부총장실
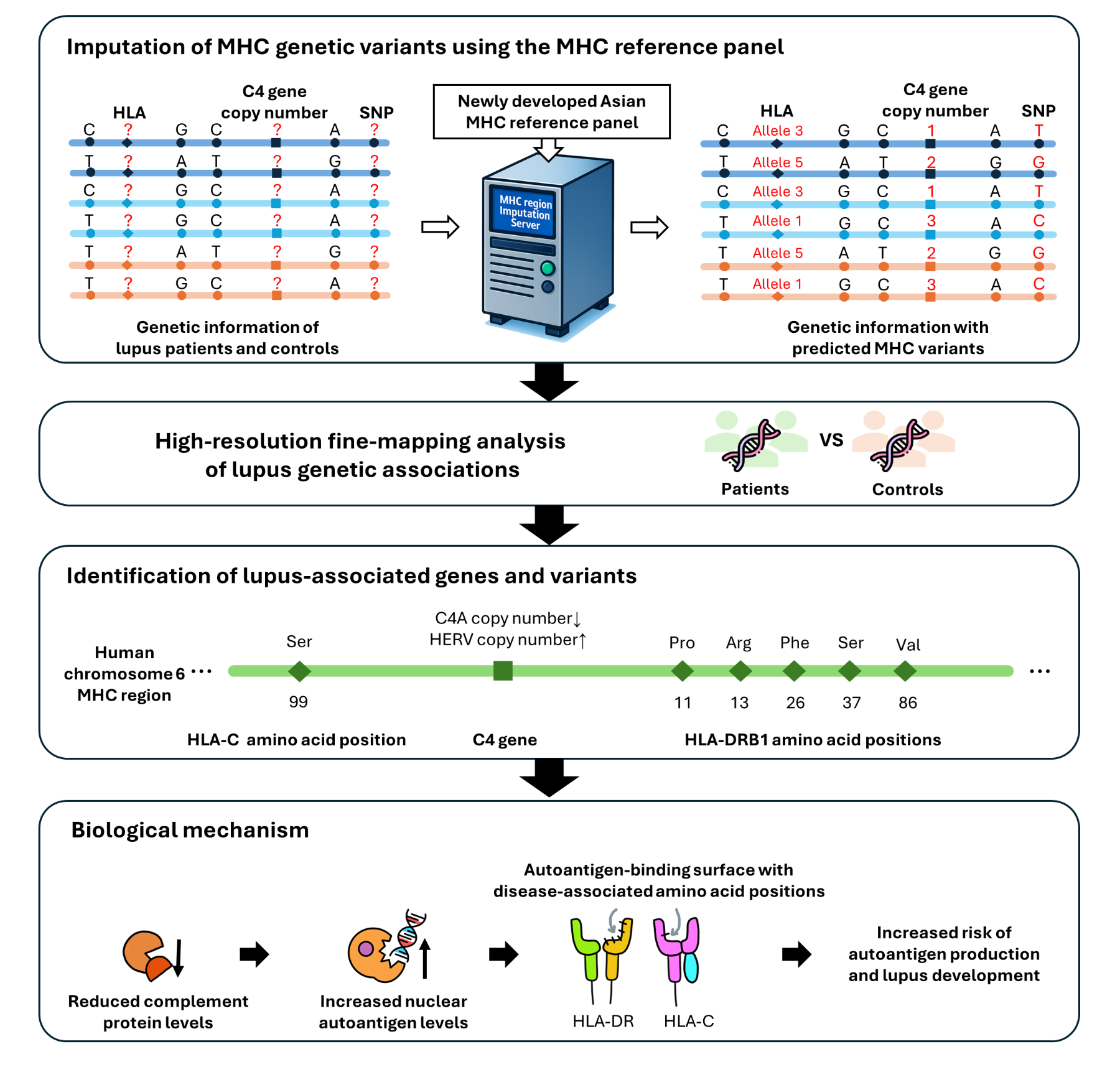
Professor Kwangwoo Kim's research team at the Department of Biology develops MHC Genetic Variation Reference Panel
Lupus-causing genetic variants identified through precise genome analysis of approximately 70,000 individuals
Systemic lupus erythematosus (SLE) is a common autoimmune disease in which the immune system mistakes healthy cells for foreign invaders and attacks them. While complex factors, including genetics, environment, and sex hormones, play a role, the exact pathogenesis remains unclear.
Amidst this, Professor Kwangwoo Kim's research team has now brought the most generically complex region of the human genome into clearer focus. By developing a high-resolution analysis of genetic information in the Major Histocompatibility Complex (MHC) region tailored to the Korean population, the team analyzed genome data from approximately 70,000 individuals and identified key genetic variants that independently and jointly shape lupus risk.
MHC region: Key to lupus genetic analysis
The MHC region of chromosome 6 is dense, polymorphic, and notoriously resistant to conventional genome-wide analysis. Its complexity has long obscured the distinct contributions of immune-related genes, particularly within the HLA cluster and the complement system. Professor Kim’s team addressed this problem at the level of infrastructure, constructing a reference panel capable of simultaneously imputing HLA amino acid residues and C4 copy number variation, which significantly improved analytical precision.
Applying this tool to genome data from approximately 70,000 individuals, including lupus patients and healthy controls, the team uncovered a stratified genetic architecture underlying SLE susceptibility. Individuals with reduced C4 gene copy numbers showed a roughly 1.4-fold higher risk of developing lupus compared to those without, while each additional copy of the gene was associated with a roughly 31% decrease in risk. Furthermore, it was discovered that specific amino acid mutations within the HLA gene alter the way the HLA protein binds to antigens, causing structural changes that can misidentify autoantigens as foreign invaders. When the C4 gene is reduced in number or an abnormally long untranslated sequence is inserted, complement protein production is reduced, increasing the likelihood of an immune system imbalance. These results demonstrate that the abnormal immune responses seen in lupus patients are intricately linked to genetic factors.
Possibility of early diagnosis and personalized treatment: laying the foundation for precision medicine
This study identified the genetic pathogenesis of lupus, establishing a foundation for precision medicine in autoimmune disease. By enabling reliable risk stratification based on specific genetic configurations, this research opens pathways toward earlier diagnosis and more individualized therapeutic strategies. It also reframes the MHC region not as an analytical obstacle, but as a tractable landscape that might hold more secrets for science and medicine.
This study was conducted jointly by Professor Kim's team at the Department of Biology, Hanyang University, the National Institute of Health, and UNIST, with support from the National Research Foundation of Korea. The results were published in the December 2025 issue of Annals of the Rheumatic Diseases (IF 20.6), a top-tier international journal in the field of rheumatic diseases, under the title, “Development of an MHC imputation panel highlights independent contributions of HLA amino acid residues and C4 copy number variations to SLE risk.”
